
Mr. PARTS
Minirhizotron Phenology and Root Traits
Start date: 06/2017 End date: 05/2019
Overview
Plant roots are a vital part of the carbon, nutrient and water cycles, but are very difficult to measure on phenological timescales as usually you have to dig them up. This is in comparison to above-ground or whole system integrated methods, such as Eddy Covariance, phenocameras or remote sensing, which can be made in continuous, long term and automatic fashions to provide time-integrated insight, Commonly, root phenology is simply assumed to be in sync with above-ground phenology, rather than directly measured. The aim of Mr.PARTS was to develop fully automated minirhizotrons systems, replicable on a reasonable experimental budget, for measuring local root phenology. This would ultimately allow a comparison at our MANIP site with above ground phenology. We also aimed to develop methods of processing these high resolution images capable of 'keeping up' with automated measurements. |

Minirhizotrons (camera systems placed within specially installed belowground observatories) are a method for non-destructive and repeatable observation of the soil system surrounding the tube, including viewing roots in undisturbed soil. The technique is limited by both the need to visit sites to make manual measurements and the need to process images for which there are few satisfactory automatic approaches. From 2D images collected from minirhizotrons, root prence can be identified, which can be used as a proxy for root biomass, as well as root traits, such as colour and morphology, which may relate to root functions (e.g. nutrient uptake potential) and vitality. Time change in these image properties gives us information about root demography and phenology.

Scientific Questions:
• How is root growth linked to nutrient status? Does root phenology change with different availability of N and P?
• How is root growth linked to water availability? Does this interact with nutrient status?
• How is does root phenology related to above-ground phenology? How does root phenology relate to ecosystem C fluxes and GPP?
• Is root growth/phenology representative of below-ground C assignment? Does nutrient status affect C used belowground in ways not measurable using minirhizotrons?
Technical Objectives

• Develop a new minirhizotron system capable of automatically collecting images up to a daily timescale
• Develop methods of image interpretation suitable for use alongside conventional human markup for high time resolution images collected from an automatic system
• Deploy these automatic systems in both a highly controlled and instrumented greenhouse mesocosm, and in the field as part of the MANIP experiment
Outcomes
• Automatic minirhizotron system: We develped 10 robotic minirhizotron systems able to collect images at timescales up to hourly. They operates automatically in the field for up to ~ 250 autonomous sampling cycles. We expect to provide more information on this system once field data is collected and processed - but feel free to get in touch if you can't wait • Mesocosm Experiment: We built a mesocosm facility with 9 Mesocosms with facilities for available for measurement of CO2 fluxes, above-ground ‘phenocam’ style camera measurements, soil moisture and root growth measurements by other methods (ingrowth cores), as well as one horizontally-deployed minirhizotron per mesocosm. In this controlled but 'realistic' setting we can look at the link between root growth and other responses, such as C fluxes and above-ground development. We are currently processing the data from this experiment for publication |

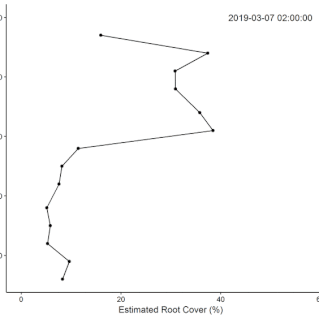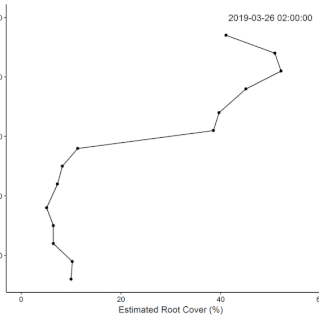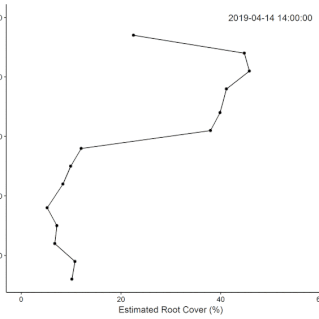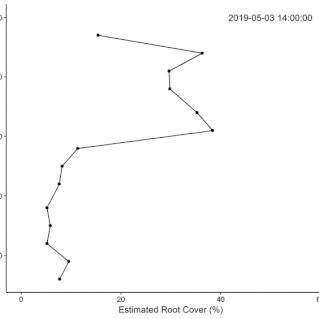
• Field Experiments (1): The technical challenge of building the system took longer than anticipated, so we did not complete the original plan for the replicated field experiment during the fellowship. Data from supplementary manual measurements were used in this
this paper showing nutrient effects on root growth varying through the (water driven) seasonal cycle.

• Image Processing: I aimed to take advantage of advances in deep learning for image analysis to interpet minirhizotron images. However we were limited by a slow pace in instrument development and the technical difficulty of the problem. We did, however, successfully develop a shallow learning method, based on gradient-boosted decision trees, for classifying root cover in images (in a similar way to the manual methods used in this paper), which allowed a substantial reduction in the man-hours needed for processing the manually collected images from our site. This method is not suitable for time series data because it needs to be re-trained on a dataset for individual soil conditions at the point of sampling. However, in future we expect to be able to employ a neural network based method (either off-the-shelf or with our own adaptions) for automated data on a continuous timescale when we have sufficient training data available.

• Field Experiments (2): Following the conclusion of MrPARTS, we continue to run the automatic minirhizotrons we have created as part of long-term measurements to unravel the link between root phenology and ecosystem C fluxes
• Tracer Experiment: I also conducted a 15N tracer experiment over the period of this experiment. This aimed to quantify the degree to which root turnover mattered to herbaceous layer plant growth by measuring how much litter N was recovered in new plant biomass. I found that the nutrient treatments affected this, implying the N cycle at the site is tighter under the more fertile NP addition (due to leaching of N, this site in the MANIP may now have excess P availability). You can read the paper here!






